MK | Ware › Solutions › Batch Traceability
Batch Traceability – Creating Transparency with Seamless Tracking
Batch tracing is essential for many companies and an important prerequisite for monitoring and guaranteeing safety and quality standards. This measure is also extremely important within a supply chain.
By labelling product deliveries with batch identification, it is possible to track exactly at which point in the supply chain products from certain production batches or lots are currently located. This means that quality checks or product recalls, for example, can be carried out effectively.
What is Batch Traceability?
Batch tracing is the tracing of the origin of a batch based on information about the components used to manufacture this batch. This ensures that for each batch it is possible to trace exactly where these components came from and how they ended up in the product. Batch traceability is an important tool for ensuring the quality and safety of products.
Goals of Batch Traceability
- Creating transparency across the entire manufacturing process
- Identifying suppliers more quickly
- Correcting errors in production more quickly
- Removing defective products from the market quickly
- Reducing the risk of product recalls and quality problems
Batch Obligation for Companies: Legal Basis
Transparent batch traceability is mandatory in many industries. The exact legal requirements vary depending on the industry and region. Companies should therefore familiarize themselves with the specific laws and standards in their industry to ensure that they meet the traceability requirements.
The industries with mandatory batches include
- Food and animal feed industry
- Pharmaceuticals and chemicals
- Healthcare and medical devices
- Automotive, railroad and aircraft industries
- Cosmetics industry
Various laws and directives stipulate batch traceability. In the food industry, for example, this is Regulation No. 178/2002, Article 18 of the European Court of Justice, in the pharmaceutical industry the Good Manufacturing Practices (GMP), and for medical technology the Medical Device Regulation (MDR). In many industries, DIN ISO 9001 is also used as a standard for quality assurance and batch traceability.
If a quality defect is detected during a production process, production must be halted until proof can be provided that the defect has been rectified. Affected products must be withdrawn from the supply chain, e.g. by recalling deliveries or issuing calls for replacement to end customers. The defective products are usually destroyed by the manufacturer.
For bulk products and goods in vibratory feeders, the batch management requirement is suspended. Here, batch inaccuracy is conceded, since deliveries cannot be separated without excessive effort due to the processing and the nature of the goods. For such products, in case of a quality issue, the entire production lot may have to be suspended.
Certain information on the origin and production process of products is also mandatory under most legal regulations. This information must be provided clearly for each batch and must be able to be provided to supervisory authorities within 24 hours.
Mandatory Information for Batch Traceability
- Batch number, Lot Number, or other Identifiable Number
- Information of Origin and Contact Details of the Manufacturer or Supplier
- Date of Production, Production Time if applicable
- Raw Materials and Ingredients used, individual Components
- Volume/quantity of Products in the Batch
- Production Equipment used
- Distribution Channels
How does Batch Traceability Work?
In order to be able to trace the exact batch of products seamlessly and transparently, they must be clearly identifiable. By uniquely marking each production batch or lot with batch numbers, companies can monitor the whereabouts of their products in the distribution network.
The production batch is noted on each precast container at the producer, each sub-packaging container of the delivery, and the delivery bill. This enables producers, suppliers and distributors to respond quickly in the event of recalls or quality issues. The batch numbers serve as an identification feature that allows affected batches to be precisely located and identified.
Downstream tracing and Upstream Tracing are two different batch traceability approaches used in the supply chain. The main difference between the two is the direction in which the tracing is performed.
Downstream-Tracing
For downstream tracing, the path of a product delivery is tracked from production through intermediaries to the end consumer. This involves capturing information about production batches and their movement as they move along the sales flow. This approach enables companies to monitor the whereabouts of their products in the distribution network and respond quickly to recalls or quality issues.
Upstream-Tracing
In contrast, upstream tracing focuses on the path of raw materials and components that go into the production of a product. Here, information about suppliers and their batches is recorded to verify the origin and quality of the materials used. As a result, the company can ensure the traceability of its products back to the suppliers and identify potential risks in the supply chain.
Both Tracing Models can also be combined to ensure comprehensive Traceability.
Regardless of which model is chosen and whether batch numbers or other identifiers are used, the batch tracing process is usually as follows.
Batch Traceability Process
- Recording information: First, all relevant information on the raw materials used, production batches and distribution channels is recorded and documented. The legally required information is thus provided.
- Labeling batches: Each production batch is given a unique identification, such as a batch number. This identification is noted on product containers and packaging.
- Documenting movements: Throughout the process, all movements of the product batch are accurately logged, for example through internal pick slips and outbound delivery slips. This includes transport from one location to another, storage and distribution. Während des gesamten Prozesses werden alle Bewegungen der Produktcharge genau dokumentiert, z. B. durch interne Entnahmescheine und ausgehende Lieferscheine. Dies umfasst den Transport von einem Standort zum anderen, die Lagerung und den Verkauf.
- Linking information: The recorded information is interconnected in order to be able to trace the entire path of the product batch. This allows quick identification of which batches are affected and where they are located in the event of a recall or quality issue.
- Guaranteeing traceability: Regular audits and reviews ensure that batch traceability is performed properly and that all information is correctly recorded and documented.
Improve the Traceability of your Products: Batch Tracing using ERP Systems
ERP Systems, i.e. Enterprise Resource Planning, make it possible to digitally schedule resources and track the completion of production steps and batches generated. Batch Traceability with ERP Systems is usually performed by linking information on individual batches to the corresponding production and supply chain processes. Here, the system is a central database where information about the batches can be retrieved.
Managing Batch Traceability with ERP systems enables companies to respond more quickly to potential problems, as batches or lots affected by errors can be identified more quickly.
Benefits of Using Batch Traceability Software with ERP Systems
Batch tracing offers many advantages for companies of all sizes. On the one hand, it creates the necessary transparency across the entire manufacturing process, which in turn facilitates quality assurance. In addition, companies can react more quickly to potential problems by tracing batches, as errors can be identified more quickly.
Overview: Advantages of Batch Tracing
- Transparency throughout the entire Manufacturing Process
- Fast Troubleshooting
- Improvement and Assurance of Product Quality
- Reliable and Retraceable Supply Chains
- Maximum Process Reliability
- Overview of all Data
How ERP Systems Work in Batch Traceability
As soon as a production order for a new batch is received, a batch number is automatically created in the system and can be tracked from then on. Batch movements are recorded from order entry to outbound: Each container of products is labeled with a barcode for accurate assignment to the batch. In addition, each individual product can be given a label with a serial number.
This barcode is scanned when goods are received and issued at each intermediate storage or production facility. In this way, every movement of the goods is tracked, and the inventory of the batch is constantly monitored.
Batch Tracing with the Software from MK|Ware
Our MK | Label software is at your side for seamless and transparent tracking of your batches. By attaching a label to a load unit, the transport and product packaging is labelled with important data that is essential for comprehensive batch traceability. This facilitates communication between the manufacturer, carrier and recipient.
In addition, our MK|Code Software can support your batch traceability processes with even more detailed insight. Your production processes can be documented with the help of individual serial numbers. This gives you a complete overview in the event of complaints. With the combination of our MK | Label and MK | Code software, you are guaranteed complete and easily assignable traceability.
How Our Batch Tracing Works
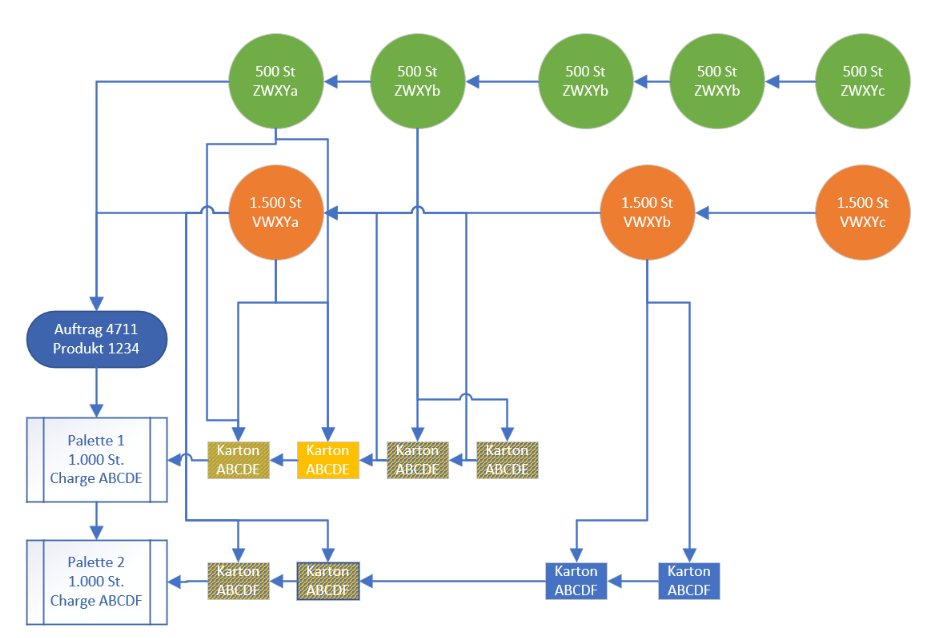
In the example shown, two primary materials (green and orange), each with a batch number, are used to produce products for an order. The products are also given batch numbers and are packed in boxes that are stored on pallets. If a production error is detected due to an inferior primary material in a box, MK | Ware can be used to identify exactly which batch of primary material is faulty. In turn, the batch can be used to identify the end products into which the input material has been incorporated, and these cartons are also blocked - the rest of production can continue while the affected cartons and materials are specifically withdrawn from circulation.
This detailed identification goes beyond the possibilities of software from other providers: without software such as MK | Label, the entire production would have to be stopped or all products written off as faulty production, as it would not be possible to trace which primary materials were used in which exact product batches.
Put your trust in MK | Ware - we will be happy to advise you on the possibilities of our solutions in your company.





